News & Articles
Browse all content by date.
By the Immunity Education Group
(http://immunityeducationgroup.org/)
Introduction by Gary G. Kohls, MD
See http://duluthreader.com/articles/categories/200_Duty_to_Warn for the two versions of this column) and see http://freepress.org/article/futilely-trying-expose-big-pharma%E2%80%99s-vaccine-cartel-facts for last week’s column that will make this one more understandable.)
The United States Centers for Disease Control & Prevention (CDC) receives about 30,000 reports of adverse vaccine reactions each year. That huge number is believed to actually represent perhaps only 1% of the actual annual vaccine-injuries. Therefore, there may actually be as many as 3 million adverse events from vaccines each year in the US.
About 2,000 of the 30,000 adverse reaction reports are officially categorized by the CDC as “serious” (ie, causing a prolonged hospital-stay, a permanent disability or a death). The CDC ignores the even more serious “chronic” consequences that commonly occur weeks or months following vaccinations that don’t result in an acute hospital stay or a sudden death and therefore don’t get listed as “serious” or even properly labelled with a diagnostic code number.
It is important to emphasize that the 30,000 reports are a gross underestimation, since physicians and clinics typically – and intentionally - fail to report to the CDC many of what they regard as “nuisance” adverse effects that are reported to them by concerned patients, parents or loved ones.
To add further to the many reasons why the CDC and the state’s departments of health adverse vaccine reaction statistics are not be trusted is that most minor, moderate, transient or chronic adverse effects may never be reported back to the clinics that did the inoculations. This phenomenon occurs partly because patients (or the parents of infants or children) have been falsely assured that any adverse effects that occur following a batch of vaccinations are to be disregarded as benign, normal and nothing to be concerned about.
Vaccine-induced Disorders are Iatrogenic Disorders
Another, more difficult to measure factor that goes unnoticed is the fact that vaccine-induced disorders are iatrogenic disorders (doctor-, drug- or surgery-caused) that, if honestly diagnosed and given proper ICD-10 code numbers, would reflect badly on Big Pharma’s thriving vaccine industry but also on those portions of the medical profession that profit from vaccinating and over-vaccinating their patients.
If such self-evident truths were ever told, there would be significant upticks in the recently-propagandized phenomenon of “vaccine-hesitancy”. The CDC doesn’t even mention iatrogenic disorders, even though one of its prime duties is about “disease prevention” not just “disease control”. My profession never used to be about making patients sick.
Honesty about what constitutes a preventable disease should be elementary, whether the disorder is of “unknown cause” or of “known causes” such as iatrogenic, infectious, nutritional, autoimmune, toxicologic, etc.
The public should be alarmed and ashamed that the (heavily pharmaceutical industry-subsidized) CDC does virtually nothing about the established epidemic of totally-preventable vaccine-induced iatrogenic disorders that are listed in the segment further below.
In addition, most clinic personnel, especially physicians and nurses, have been indoctrinated in their student training (and later in their regular “indoctrination sessions” from drug/vaccine corporate sales staff and other agents in the “healthcare” profession) to believe the “Big Lie” that all vaccines are 100% safe and 100% effective.
Therefore, those industries that profit from over-vaccination agendas often refuse to believe the “staring-them-in the-face-truths” (that are published in every vaccine [and prescription drug] “product insert”) that vaccines (and also prescription drugs) can be extremely toxic – even to the point of lethality.
Cognitive Dissonance and the Over-Vaccinating of America’s Children
Cognitive dissonance is the “emotional discomfort felt when deeply held beliefs are contradicted by new information”. It.is a potent psychological factor in the practice of industrial-strength medicine. It can be so powerful that anybody, even physicians, can go through an entire career believing profitable lies.
In the case of the over-vaccinating of infants and toddlers, entities that profit from that lucrative reality need to stay busy ignoring unwelcome facts or, when cornered, attack the facts – that is, if they are ever confronted with them., This issue is urgent and serious, because millions of innocent children are being made chronically ill every day by the common, highly profitable agenda of over-vaccinating as many infants and small children as possible.
Many are the American healthcare practitioners that have been influenced by the cunning salesmanship of the pharmaceutical industry, and thus they have been able to overcome their cognitive dissonance while simultaneously ignoring the many warning signs of America’s over-vaccination agendas, not to mention the epidemics of vaccine-induced autoimmune disorders, allergic disorders, asthma, type I childhood diabetes mellitus (an autoimmune disorder likely related to aluminum-containing vaccines), autism spectrum disorders, learning disorders, rheumatological disorders, etc. (See the supporting data in the second half of this column.)
To make matters worse, neither the inoculators nor the inoculees of vaccines appear to have read or understood the warning information that is in the “product insert” sheets that come with every vaccine vial (and prescription drug).
At best, those product inserts only list the “acute” (as opposed to the “chronic”) toxic effects that have been observed in short-term studies that were performed by the vaccine manufacturers prior to FDA (Federal Drug Administration) approval. Such short-term studies are still commonly done without regulatory agency oversight. Chronic studies of vaccine safety and efficacy are typically not done!
What Could Possibly Go Wrong with that Picture?
Therefore, vaccine-induced-but-often-delayed neurological disorders such as autism spectrum disorders, learning disorders, behavioral disorders, seizure disorders, encephalopathies, sudden infant death syndrome (SIDS), near-SIDS, etc are not acknowledged to be related to vaccines by the CDC (Centers for Disease Control and Prevention), the WHO (World Health Organization), the AAP (American Academy of Pediatrics) and every state department of health.
Likewise, vaccine-induced autoimmune disorders such as juvenile type 1 diabetes mellitus, allergies, asthma, macrophagic myofasciitis, the Autoimmune/Inflammatory Syndrome Induced by Adjuvants (ASIA), lupus, rheumatoid arthritis, infant mitochondrial disorders, non-polio transverse myelitis, Guillain-Barre Syndrome, multiple sclerosis, menstrual disorders, infertility (the latter two commonly occurring in Gardasil recipients), etc, are often left out of CDC, WHO, and AAP documents.
Another serious caveat that the parents of infants scheduled to receive cocktails of vaccines need to know about concerning CDC statistics is that there have never been done any lab animal studies (much less human studies) that have assessed the safety of two or more vaccines being injected simultaneously - which is common practice in America as well as in most “advanced” first world countries.
“Toxic Synergism” and its Importance in this Discussion
There is a scientific truism that states that two toxins administered simultaneously often have “synergistic” adverse toxic effects (as opposed to “additive” effects). What synergism means in terms of vaccine toxicity is that when two or more toxins are administered simultaneously, the incidence and severity of their adverse effects can often be multiplied, rather than simply added!
Here is an example of this principle: If a solution of mercury that is known to kill 1% of a group of lab animals (known as the Lethal Dose 1% or LD1 for mercury) is injected into the animals simultaneous with a solution of lead that is known to kill 1% of the lab animals (the LD1 for lead), 100% of the lab animals will die.
One would have expected that 2% of the lab animals would have died if toxicities were simply additive. That principle likely accounts for the reality that over-vaccinated babies have such a huge variety of toxicities (see above and below) and also that there are statistical projections for the incidence of vaccine-related autism spectrum disorders in the United States (estimated to approach 50% for boys by the year 2030 - if vaccine policy is not changed in the US). See the chart projection below:
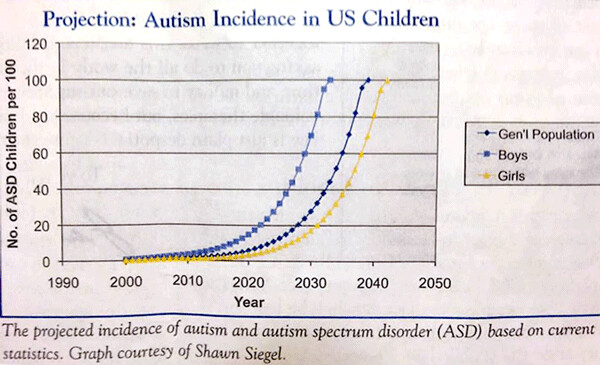
It is also well-established that the presence of injectable aluminum adjuvants in vaccines is a common cause of autoimmune disorders. But aluminum in vaccines is also a potent neurotoxin. Aluminum adjuvants, as I have frequently mentioned in this column, has been added to many infant vaccines because the metal, when incubated with proteinaceous antigenic tissue (both intended and inadvertent), has been known to cause a far-more potent immune response than when viruses and other antigens were injected into muscle tissue without it. Most of the new vaccines that were added to the infant vaccine regimen since Thimerosal (mercury) was essentially banned have contained aluminum.
Below is an abstract from a 2011 medical journal article that warns about the adverse health consequences of the increasingly common inclusion of aluminum adjuvants in infant vaccines: Curr Med Chem. 2011;18(17):2630-7
Aluminum Vaccine Adjuvants: Are They Safe?
Tomljenovic L; Shaw CA https://www.ncbi.nlm.nih.gov/pubmed/21568886 Abstract
“Aluminum is an experimentally demonstrated neurotoxin and the most commonly-used vaccine adjuvant. Despite almost 90 years of widespread use of aluminum adjuvants, medical science's understanding about their mechanisms of action is still remarkably poor. There is also a concerning scarcity of data on toxicology and pharmacokinetics of these compounds. “In spite of this, the notion that aluminum in vaccines is safe appears to be widely accepted. Experimental research, however, clearly shows that aluminum adjuvants have a potential to induce serious immunological disorders in humans.
“In particular, aluminum in adjuvant form carries a risk for autoimmunity, long-term brain inflammation (encephalitis) and associated neurological complications and may thus have profound and widespread adverse health consequences.
“In our opinion, the possibility that vaccine benefits may have been overrated and the risk of potential adverse effects underestimated, has not been rigorously evaluated in the medical and scientific community.
“We hope that the present paper will provide a framework for a much needed and long overdue assessment of this highly contentious medical issue.”
Mercury (in the form of the potent neurotoxin Thimerosal) was commonly used as a cheap anti-bacterial preservative in most multiple-dose vaccine vials – especially during the dramatic autism epidemic of the late 1980s and 1990s - until concerned pediatrician-members of the American Academy of Pediatrics accurately saw the connections and pleaded with vaccine manufacturers (and convicted felons) such as Pfizer, GlaxoSmithKline, Eli Lilly and Merck to stop putting the neurotoxin thimerosal in its infant vaccines.
Caring pediatricians back then listened to parents and therefore KNEW that the over-vaccination of infants with mercury-containing vaccines was causing an escalating variety of neurologic/emotional/learning/behavioral disorders – only one small (albeit dramatic) example of which were the autistic spectrum disorders. It was obvious to many parents, lay observers and even professionals that fully-immunized children were far sicker – both acutely and chronically - than the so-called “under-vaccinated”.
By the year 2000, the vaccine manufacturers finally took mercury out of most vaccines (except for the influenza vaccines) - but only after they had used-up or unloaded their stock-piled supply of mercury-containing vaccines to unsuspecting clinics around the world! But by that time, new vaccines with a multitude of new toxic substances (in addition to aluminum) were being rapidly added to the CDC- and AAP-recommended schedules. None of the new toxic substances have ever been thoroughly tested, especially for synergistic toxicity.
What Could Possibly go Wrong?
What is being discussed above needs repeating:
There is no known safe dose of any combination of either of the two most toxic vaccine-additives (mercury and aluminum) when they are given to animals, especially if they are administered intramuscularly. But when two or more neurotoxins are injected simultaneously, far more toxicity occurs than if the toxins had been administered at long intervals.
The following lists of adverse effects and the unavoidably toxic ingredients in vaccines are necessarily incomplete. The lists are mostly obtained from the vaccine manufacturer’s FDA-approved product insert lists and/or from the Vaccine Information Statements of the CDC.
(The bolding and italicizations in the article below are by the Duty to Warn author.)
Please re-read the important Duty to Warn column from 5-15-2019 (and a number of other anti-over-vaccination articles that have been posted) at: http://duluthreader.com/articles/2019/05/16/16975_futilely_trying_to_expose_big_pharmas_vaccine-55294-1
Vaccine Side Effects & Ingredients
By the Immunity Education Group
http://immunityeducationgroup.org/vaccinesideeffectsingredients/
1. Hepatitis B Vaccine The CDC recommends all infants receive 3 doses of hepatitis B vaccine starting on the day of birth.
Reported side effects include: fever, poor feeding, lethargy, irritability, headache, fatigue, joint and body aches, anaphylactic allergic reactions, worsening of Multiple Sclerosis symptoms, vascular inflammation, onset of Lupus and other autoimmune disorders, Stevens-Johnson Syndrome (a severe allergic reaction involving skin and internal organs), heart palpitations, asthma attacks, minor liver damage, bleeding from low platelets counts, eczema, bruising, hair loss, eye inflammation, vertigo, ringing in the ears, migraines, nerve and muscle weakness or paralysis, Guillain-Barré syndrome, spinal cord inflammation, seizures, optic nerve inflammation, and multiple sclerosis.
Ingredients in the vaccine include: Hepatitis B surface antigen (the proteins on the outer shell of the virus), aluminum (250 micrograms), saline, yeast proteins, phosphate, and formaldehyde (depending on brand).
*All 3 doses are now mandated for Kindergarten school entry in California. The brand names are Recombivax HB and Engerix-B. Hepatitis B vaccine is also in the combination brand Pediarix.
2. DTaP Vaccine The CDC recommends 5 doses of DTaP vaccine (Diphtheria, Tetanus, acellular Pertussis) are given between 2 months and 5 years of age. A 6th dose of this vaccine is recommended at age 12 as the Tdap vaccine.
Reported side effects include: pain, redness, swelling, or a nodule at the injection site, fever, poor appetite, sleepiness, vomiting, diarrhea, headache, body aches, rash, non-stop crying for three or more hours (indicates encephalitis), febrile seizures, grand mal seizures, long-term seizures, fever greater than 105 degrees, anaphylactic shock, brain injury, coma, Guillain-Barré syndrome, brachial neuritis (dysfunction of the nerves in the arm), cyanosis, extensive swelling of the injected limb and nearby joints, cellulitis (skin infection), abscess, hypotonic-hyporesponsive episode (shock), hypotonia, bronchitis, pneumonia, lymph node swelling, bleeding from low platelet counts, encephalopathy (permanent brain damage), apnea, and Sudden Infant Death Syndrome.
Additional reactions reported after the Tdap vaccines include nausea, abdominal pain, severe migraine, nerve compression in the neck, diabetes, heart inflammation, facial nerve dysfunction, inflammation of the spinal cord, and Henoch-Schonlein purpura (severe bruising and kidney dysfunction).
Ingredients in the DTaP vaccine include: proteins and toxin elements from the pertussis germs, toxin components from tetanus and diphtheria, aluminum (between 330 and 650 micrograms, depending on the brand), saline, formaldehyde, glutaraldehyde, 2-phenoxyethanol, and polysorbate 80. Brand names are Infanrix, Daptacel, and Tripedia. DTaP vaccine is also in the combination brands Pediarix, Pentacel, and Quadracel. Tdap brand names are Adacel and Boostrix. *Some plain Tetanus vaccines and DT/Td vaccines (Diphtheria and Tetanus, without Pertussis also contain mercury.
3. Hib Vaccine The CDC recommends 4 doses of Hib vaccine between 2 and 15 months.
Reported side effects of Hib include: redness, swelling, and pain at the injection site, high fevers, swollen lymph nodes, anaphylactic shock, febrile seizures, abscess, extensive swelling of the injected limb, grand mal seizures, hypotonic-hyporesponsive epidose (shock), sleepiness, fainting, apnea, hives, and rash.
Ingredients in the Hib vaccine include: sugars from the Hib germs bonded to either tetanus toxoid or meningococcal proteins (to help induce a better immune response), aluminum (225 micrograms in the Merck brand only), saline, sucrose or lactose sugar, and formaldehyde. Brand names are ActHIB, PedvaxHIB, and Hiberix. Hib vaccine is also in the combination Pediacel.
4. Pneumococcal Vaccine The CDC recommends 4 doses of PC vaccine between 2 and 15 months.
Reported side effects include: fever, poor appetite, sleepiness, vomiting, diarrhea, headache, body aches, rash, pain and swelling at the injection site in about 50% of people. Fussiness or irritability are reported in 80%. Other side effects include pneumonia, wheezing, severe allergic reactions, febrile seizure lymph node swelling, anaphylactic shock, angioneurotic edema (severe, painful swelling), and apnea.
Ingredients in the vaccine include: sugars from the outer shell of the germ bonded to diphtheria toxoid proteins, aluminum (125 micrograms), polysorbate 80, and succinate. Brand names are Prevnar and Prevnar 13.
5. Rotavirus Vaccine The CDC recommends the oral liquid Rotavirus vaccine in 2 or 3 doses between 2 and 6 months (depending on the brand).
Reported side effects include: fever, vomiting, diarrhea, seizure, poor feeding, irritability, Kawasaki disease (a severe autoimmune reaction that affects the immune and cardiovascular systems), intussusception (severe intestinal blockage that may require emergency surgery), bloody stools, bleeding problems from low platelet counts, and hives.
Ingredients in the vaccine vary significantly between the two brands: Merck’s RotaTeq vaccine includes five strains of live Rotavirus germs grown in monkey kidney cells (called VERO cells), sucrose, sodium citrate, sodium phosphate, and sodium hydroxide, polysorbate 80, traces of fetal cow blood (nourishes the VERO cells), and DNA from circovirus (a pig virus which contaminates an enzyme used in vaccine manufacturing). GSK’s Rotarix brand contains a single strain of the live virus grown in VERO cells, sucrose, dextran, sorbitol, amino acids, a solution of vitamins/minerals/phenol red, calcium carbonate, xanthum gum, water, and circovirus DNA. Brand names are Rotarix (Glaxo) and RotaTeq (Merck).
6. Polio Vaccine The CDC recommends 4 doses of Polio vaccine between 2 months and 5 years of age.
Reported side effects include: redness, swelling, pain, or mass at the injection site, allergic reaction, anaphylactic shock, fever, poor appetite, sleepiness, irritability, crying, vomiting, Guillain-Barré Syndrome, lymphadenopathy, joint and muscle aches, febrile seizures, grand mal seizures, numbness, and sleepiness.
Ingredients in the vaccine include: three strains of the virus (inactivated so it isn’t live), saline, vitamins, amino acids, sucrose, glutamate, human albumin (blood proteins filtered out of donated human blood), 2-phenoxyethanol, formaldehyde, three antibiotics, and calf serum. Brand name is IPOL. Polio vaccine is also in the combinations Pediacel and Pediarix.
7. MMR Vaccine The CDC recommends 2 doses of Measles, Mumps, Rubella combo vaccine at 1 year and 5 years of age.
Reported side effects include: measles infection, mumps infection, rubella infection, pain, swelling, and redness at the injection site, fever, rash, encephalitis, encephalopathy (permanent brain damage), fainting, headache, dizziness, body aches, irritability, vomiting, diarrhea, parotid gland swelling (in the cheeks) lymph node swelling, arthritis reaction (in about 3% of children and 12 to 26% of teen and adult women: varies from mild and temporary to permanent and severe), coma, febrile seizures, long-term seizures, subacute sclerosing panencephalitis, Guillain-Barré Syndrome, ataxia, multiple nerve dysfunction, aseptic meningitis, deafness, vision loss, testicular swelling, bleeding problems from low platelet counts, panniculitis (tender and inflamed skin nodules, fatigue, weight loss), vasculitis, inflammation of the pancreas, diabetes, elevated white blood cell counts, anaphylactic shock, hives, muscle inflammation, pneumonia, and Stevens-Johnson Syndrome (severe allergic reaction involving the skin and internal organs).
Ingredients in the vaccine include: the three live viruses (the rubella virus was harvested from an infected aborted baby back in the 1960s), saline solution, sugar, gelatin, synthetic albumin proteins (was human blood albumin until 2007), cow fetus serum, neomycin (antibiotic), chick embryo proteins, proteins and DNA from the human fetal lung cells used to nourish the viruses during manufacturing. Brand name is MMR II (Merck). MMR vaccine is also in the combination ProQuad.
8. Varicella (chicken pox) Vaccine The CDC recommends 2 doses of chicken pox vaccine at 1 and 5 years of age. (It is also given to the elderly as the shingles vaccine).
Reported side effects include: pain, swelling, and redness at the injection site, fever, febrile seizures, bleeding from low platelet counts, chicken pox-like rash, chicken pox infection, body aches, irritability, fatigue, intestinal symptoms, headache, pneumonitis (lung inflammation), encephalitis, stroke, spinal cord, inflammation and dysfunction, Guillain-Barré Syndrome, facial nerve paralysis, dizziness, loss of coordination, grand mal seizures, meningitis, pneumonia, Stevens-Johnson syndrome, Henoch-Schonlein purpura, bacterial skin and tissue infections, and shingles. An additional side effect reported during initial safety studies of the shingles vaccine in the elderly is a 10 to 25% higher rate of heart failure or heart attack within one month of vaccination compared to placebo.
Ingredients in the vaccine include: the live virus, sucrose, saline, gelatin, MSG, potassium, DNA and proteins from the human fetal cells used to nourish the virus during manufacturing, EDTA, Neomycin, and fetal cow serum. *The combination MMR/Chicken pox vaccine (MMRV – ProQuad, by Merck) lists two ingredient changes which vary from the individual vaccines: 1. The combo shot contains ten times the amount of chicken pox virus than the separate formulations, 2. The combo utilizes human blood albumin instead of synthetic albumin. Brand name is Varivax. Varicella vaccine is also in the combination ProQuad.
9. Flu Vaccine The CDC recommends yearly doses of the flu vaccine for every person every year starting at 6 months of age. Reported side effects include: flu-like symptoms, pain, redness, and swelling at the injection site, irritability, fever, febrile seizures, Guillain-Barré Syndrome, drowsiness, loss of appetite, vomiting, diarrhea, allergic reactions, anaphylactic shock, bleeding from low platelet counts, swollen lymph nodes, inflammation of the brain and spinal cord, encephalopathy, nerve dysfunction in the eyes, face, or arm, fainting, dizziness, inflammation of the blood vessels, shortness of breath, Stevens-Johnson syndrome, chest pain, body aches, fatigue, rapid heart rate, eye infection, eye redness and swelling, tonsillitis, asthma flare up, Henoch-Schonlein purpura, difficulty swallowing, muscle tightness, abnormal gait, arthritis, tremors, and limb paralysis. (There are over a dozen different brands of injected flu vaccine and detailing exact ingredients for each is beyond the scope of this section).
The ingredients most commonly utilized in the majority of flu vaccines include: 3 to 4 strains of flu viruses which are killed and split open, saline, egg proteins, formaldehyde, mercury (in about half of the flu vaccines, those that come in a 10-dose vial), and antibiotics. In addition, every flu vaccine contains three or four of the following chemicals: octoxynol-10, Polysorbate 80, Polysorbate 20, sodium deoxycholate, octylphenol ethoxylate, betapropiolactone, nonylphenol ethoxylate, cetyltrimethylammonium bromide, sodium taurodeoxycholate, MF59 (a squalene-based adjuvant), sorbitan trioleate, barium, and Triton X-100. The live-virus nasal spray contains 4 strains of live flu virus, egg, MSG, pig gelatin, amino acids, potassium sucrose, and an antibiotic. Brand names are Fluzone, Fluarix, FluLaval, FluMist, Afluria, Agriflu, Fluad, Flublok, Flucelvax, and Fluvirin.
10. Hepatitis A Vaccine The CDC recommends 2 doses of Hep A vaccine for all babies at 12 and 18 months of age.
Reported side effects include:pain, swelling, and redness at the injection site, fever, fatigue, loss of appetite, headache, seizures, bleeding problems from low platelet counts, Guillain-Barré Syndrome, encephalitis, difficulty walking, dizziness, anaphylactic shock, allergic reactions, encephalopathy, multiple sclerosis, various nerve dysfunctions, spinal cord inflammation, fainting, vasculitis, shortness of breath, hepatitis, jaundice, rashes, flu-like symptoms, and muscle stiffness.
Ingredients in the vaccine include: the hep A viruses (whole but killed), aluminum (225 micrograms), saline, proteins and DNA from human fetal cells, cow blood proteins, formaldehyde, sodium borate, 2-phenoxyethanol, amino acids, Polysorbate 20, and antibiotics. Brand names are Havrix and Vaqta.
11. Meningococcal Vaccine The CDC recommends 2 doses of Meningococcal vaccine at 12 and 16 years of age for all children.
Reported side effects include: pain, swelling, and redness at the injection, headache, fatigue, facial nerve dysfunction, spinal cord inflammation and dysfunction, general ill feeling, joint aches, drowsiness, irritability, loss of appetite, fever, Guillain-Barré Syndrome, anaphylactic shock, allergic reactions, fainting, encephalitis, and muscle aches.
Ingredients in the vaccine include: sugars from the outer shell of the meningococcus germ bonded to diphtheria toxin, saline, and formaldehyde. Brand names are Menactra, Menomune, and Menveo. 12. Human Papillomavirus (HPV) Vaccine The CDC recommends 3 doses of HPV vaccine at 11 or 12 years of age.
Reported side effects include: pain, redness, and swelling in most people (severe in about 10%), headache, fatigue, muscle aches, joint pain, premature menopause in young adult women, nausea, vomiting, abdominal pain, fever, hives, fainting, numerous autoimmune reactions (such as autoimmune arthritis, thyroiditis, optic nerve and eye inflammation, inflammatory bowel disease, autoimmune hemolytic anemia, and multiple sclerosis), birth defects (if given while pregnant), bleeding from low platelet counts, lymph glad swelling, pulmonary embolism, pancreatitis, chills, fatigue, mild allergic reactions, anaphylactic shock, joint and muscle aches, encephalitis, Guillain-Barré Syndrome, nerve dysfunction, paralysis, seizures, fainting with seizures, spinal cord inflammation and dysfunction, severe swelling of a limb, and blood clots in the limbs.
Ingredients in the vaccine vary significantly between the two brands: Gardasil (Merck) contains proteins from the outer shell of 4 strains of HPV, aluminum (225 micrograms), saline, amino acids, Polysorbate 80, sodium borate, yeast proteins, and water.
*The updated version of Gardasil, called Gardasil 9, has the same ingredients except that it contains 9 strains of HPV proteins and has twice the amount of aluminum (500 micrograms). *Cervarix brand (GlaxoSmithKline) contains proteins from two strains of HPV, 173 micrograms of aluminum, 3-O-desacyl-4′-monophosphoryl lipid A, saline, sodium dihydrogen phosphate dehydrate, insect cell proteins, and baculovirus proteins (used in manufacturing). Brand names are Gardasil, Gardasil 9, and Cervarix.
Sources:
Click here to review the CDC Vaccine Information Statements for yourself: http://www.cdc.gov/vaccines/hcp/vis/index.html Click here to view FDA Vaccine Product Inserts
Dr Kohls is a retired family physician from Duluth, MN, USA. Since his retirement from his holistic mental health practice he has been writing his weekly Duty to Warn column for the Duluth Reader, northeast Minnesota’s alternative newsweekly magazine. His columns, which are re-published around the world, deal with the dangers of American fascism, corporatism, militarization, racism, xenophobia, malnutrition, sea level rise, global warming, geo-engineering, solar radiation management, Big Copper Mining’s conscienceless exploitation of northeast Minnesota’s water-rich environment, Big Medicine’s over-screening, over-diagnosing, over-treating, Big Pharma’s over-drugging and Big Vaccine’s over-vaccination agendas (particularly of tiny infants), as well as other movements that threaten human health, the environment, democracy, civility and the sustainability of life on earth. Many of his columns have been archived at a number of websites, including these four: http://duluthreader.com/search?search_term=Duty+to+Warn&p=2; http://www.globalresearch.ca/author/gary-g-kohls; http://freepress.org/geographic-scope/national; and https://www.transcend.org/tms/search/?q=gary+kohls+articles
NOTE: The views expressed here are those of the author and do not necessarily represent or reflect the views of Reader Weekly.”
| Tweet |


